What will health care look like 20 years in the future?

What will health care look like 20 years in the future?

2000
Gavi launches
Gavi, the Vaccine Alliance, launches to expand global access to vaccines for children in lower-income countries. Since 2000, Gavi and partners have helped immunize more than 1 billion children and prevent more than 18.8 million future deaths.

2002
The Global Fund
The Global Fund to Fight AIDS, Tuberculosis and Malaria launches and quickly becomes the world’s largest funder of programs to combat the three diseases.
Since its inception, the fund has invested more than US$60 billion, helping to save more than 59 million lives.
2003
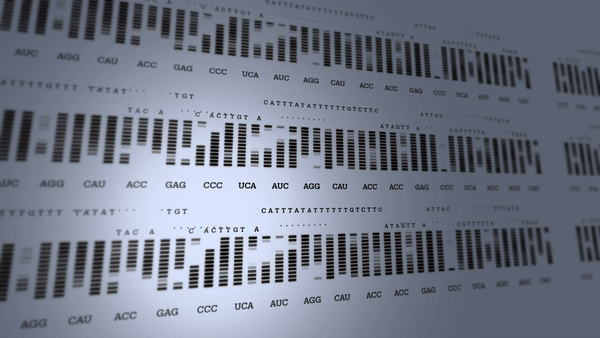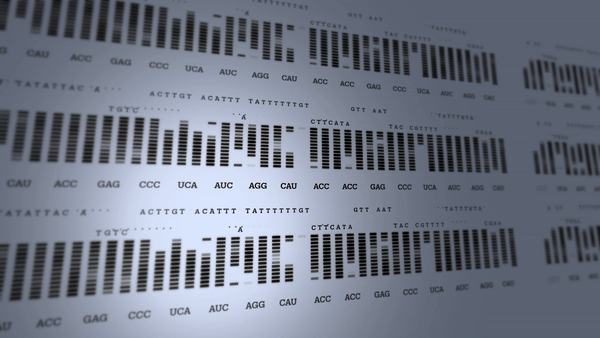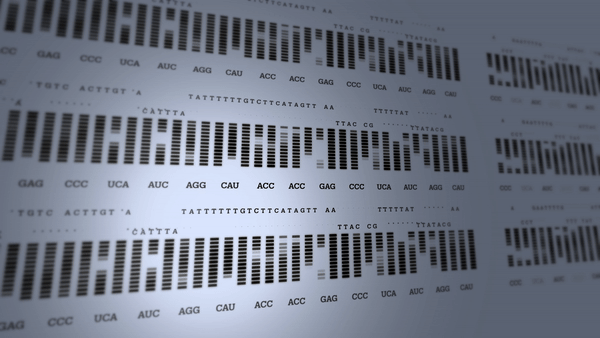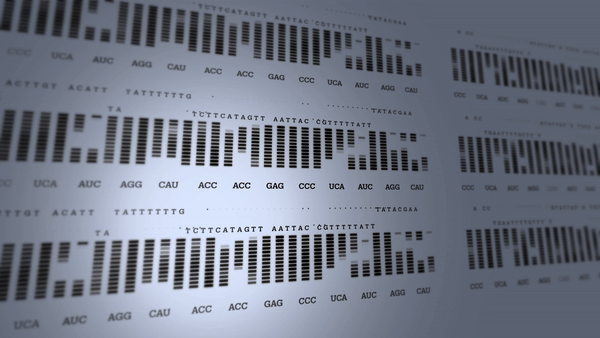
First human genome mapped
The Human Genome Project begins to revolutionize research into the diagnosis and treatment of many diseases.

2003
PEPFAR launches
U.S. President George W. Bush launches the United States President’s Emergency Plan for AIDS Relief (PEPFAR).
Since its inception, PEPFAR has helped save more than 25 million lives and enabled more than 5.5 million babies to be born HIV-free.

2006
HPV vaccine
The U.S. Food and Drug Administration (FDA) approves the world’s first vaccine against human papillomavirus (HPV). HPV vaccines are effective in preventing up to 90% of cervical cancer cases. Cervical cancer is the fourth most common cancer affecting women globally, with 90% of deaths occurring in low- and middle-income countries, where access to HPV vaccines, screening, and treatment remain limited.
In 2023, results from the KENya Single-dose HPV-vaccine Efficacy (KEN SHE) study are published, showing that a single dose of the HPV vaccine is as effective as a two- or three-dose regimen in preventing HPV infection.

2006
Rotavirus vaccination
Gavi adds vaccines against rotavirus, the most common cause of diarrheal deaths among children under age 5, to its programs.
In 2009, the World Health Organization (WHO) recommends rotavirus vaccines for all national vaccination programs, paving the way to greater access in low-income countries, where most rotavirus deaths occur.

2009
Pneumococcal vaccine
Gavi launches an innovative financing mechanism to accelerate the global rollout of the pneumococcal conjugate vaccine (PCV), which helps protect against pneumonia, the leading infectious cause of death in children.
In 2019, WHO prequalifies a new PCV developed by Serum Institute of India and PATH, making this lifesaving vaccine more affordable for lower-income countries.

2010
First rapid molecular test for TB
The first rapid molecular test for the detection of tuberculosis (TB) is released after decades of reliance on sputum-smear microscopy. The new test helps diagnose TB more accurately and identify drug resistance earlier.

2012
Truvada PrEP approved
Truvada, a combination therapy designed to provide preexposure prophylaxis for HIV-negative individuals at high risk of infection, is approved by the FDA, ushering in a new era in HIV prevention.

2015
CRISPR
A revolutionary technique for precisely editing DNA opens up entirely new approaches to improving health, from altering mosquitoes’ ability to transmit malaria to possible cures for human genetic diseases.
Watch to learn more about CRISPR

2017
CEPI launches
The Coalition for Epidemic Preparedness Innovations (CEPI) is launched to accelerate the development of vaccines against epidemic threats and expand access to lifesaving vaccines.

2021
COVID-19 mRNA vaccines
In just 10 months, scientists develop safe and effective COVID-19 vaccines using mRNA technology. Decades of foundational research into mRNA allowed these vaccines to be produced quickly, and the technology is now being explored for the prevention of many other deadly diseases.

2021
WHO recommends first-ever malaria vaccine
The long-awaited malaria vaccine is a remarkable technical achievement that, alongside other tools, including bed nets, SMC, and indoor residual spraying, could save tens of thousands of young lives each year and inform the development of next-generation vaccines against malaria. The vaccine, called RTS,S/AS01, is also the first vaccine to combat a human parasite.

2021
HIV drug
FDA approval of cabotegravir, an injectable drug for HIV prevention, adds another option to a range of treatments that have reduced deaths from HIV/AIDS and related illnesses by 69% over 20 years, from 2 million in 2004 to 630,000 in 2022.

2023
Dual insecticide-treated bed nets
WHO recommends two new types of bed nets that are treated with two insecticides, to help countries overcome growing mosquito resistance to commonly used insecticides. Evidence from a trial in Tanzania shows that these nets nearly halve cases of malaria compared to pyrethroid-only bed nets.
Discover the next generation of healthcare technology
New diagnostic tests, treatments, and vaccines in the research and development pipeline promise to be cheaper, more effective, and easier to deliver.
Imagine what’s possible in the next 20 years if the world keeps prioritizing health innovation for everyone.
Health innovations on the horizon
Discover more on the power of science, technology, and innovation
Read next





